To develop and implement the optimal CT lung cancer early detection and screening strategy within Europe
In Europe, there are 470,000 new diagnoses of lung cancer and over 380,000 lung cancer deaths every year [1]. This makes lung cancer the most lethal form of cancer. However, implementing the current NELSON protocol through national screening programmes could potentially save 60,000-80,000 lives. And this is only within Europe
One of the reasons why lung cancer has such a high mortality rate, is due to the fact that diagnosis typically occurs when the cancer is in its later stages of development. More than 50% of all lung cancer cases are diagnosed in stage IV, where 5-year survival rates (the percentage of patients still alive five years after diagnosis) range from 2% to 13% [2].
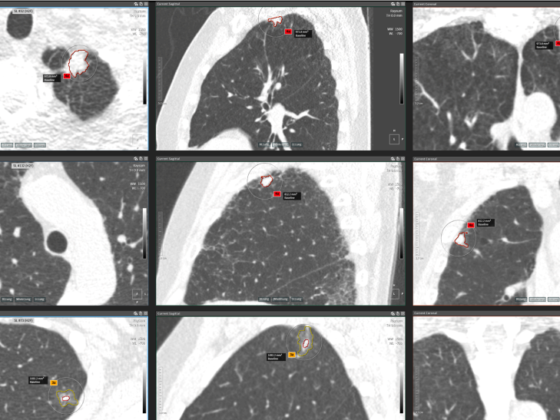
Screening for lung cancer using low-dose CT-scans has been shown to effectively detect lung cancer in the earliest stages, with more than 75% of newly detected tumours within that specific stage [3]. The NELSON study showed that this could potentially result in a lung cancer mortality reduction of 24% in males [4] and up to 59% in females [5].
Want to know more? Click here
Safe and cost-effective implementation
The 4-In-The-Lung-Run (4ITLR) trial, funded by the European Committee’s Horizon2020 programme, will investigate the most safe and cost-effective methods how to implement the NELSON early detection protocol. When successful, the study will lead to established and risk-based screening strategies which have demonstrated to be effective, affordable, acceptable to the people, cost-effective and suitable for implementation. iDNA is the key partner within the project, responsible for imaging data acquisition, data image analysis and quality control.
More specifically, together with a consortium of leading European institutions, headed by Erasmus MC and iDNA, we will develop and implement the optimal country specific and personalized CT lung cancer screening programme for high-risk populations within five European countries (The Netherlands, Germany, France, Italy and Spain). In this trial, participants with a history of intense smoking, will receive a low-dose CT-scan (LDCT) of the chest. These scans can detect small nodules in the lungs during the earliest stages of lung cancer. This means the required treatment can now be initiated in the earliest stages, before the tumour spreads, which results in a substantial increase in the survival rate of the screened participants.
Different screening approaches
In the 4ITLR trial, the consortium will investigate the effect of a screening interval of two years after a negative baseline scan. In a negative baseline scan no abnormalities are found and the NELSON study has shown that in this group the chances of developing a malignant lung cancer are much lower than people who encountered a positive baseline scan. The consortium will validate this by randomizing participants after baseline in an annual or biennial group. If proven, this will save a significant number of annual scans (up to 60%), thereby reducing unnecessary costs. This could remove important hurdles in moving forward to implement lung cancer early detection programmes.
Furthermore, the consortium will include comorbidities to lung cancer as well. These include Coronary Heart Diseases (CHD) and COPD. These diseases strongly coincide with lung cancer most likely because they share the same risk profile due to heavy smoking. In the study, lung cancer, as well as CHD and COPD, will be registered in the same CT examination.
The 4ITLR will look into the most cost-effective recruitment methods for the (hard-to-reach) target population. The consortium will do this by using a tailored recruitment approach targeting specific groups by combining social media and more traditional methods.
Finally, the consortium will look into gender differences and connecting smoking cessation programmes to early detection programmes.
4ITLR drives innovation forward
There are currently many academic institutions and companies determined to reduce lung cancer mortality. They all posses extensive, in-depth knowledge and expertise required to achieve this goal. In addition, they are ready to invest and explore new possibilities, methods, products and services.
Low-dose CT data, for example, used to diagnose lung cancer at an early stage can also be accompanied with other additional diagnostic tooling, such as using circulating tumour DNA. Combining these biomarkers with CT imaging data may increase its diagnostic potential. Next to lung cancer, this could also apply to detect CHD and COPD.
Another example is the potential of Artificial Intelligence. Saving time, preventing human error and leaving the Radiologist with only relevant imaging data to asses. Therefore, AI has the potential to make lung cancer early detection and screening programmes much more efficient.
No early detection programme will lead to results without proper and timely treatment. Examples include the latest state-of-the-art and minimal invasive instruments or by using tumour ablation technologies. All with the aim to treat the tumour with minimal discomfort to the patient. This will increase the chances of survival, especially after lung cancer is being diagnosed in an early stage (stageshift). Patients diagnosed with the earliest stages of lung cancer, may also benefit from (early) drug treatment.
- Ferlay J, Colombet M, Soerjomataram I, et al. Cancer incidence and mortality patterns in Europe: Estimates for 40 countries and 25 major cancers in 2018. Eur J Cancer 2018; 103: 356-87
- Sources: World Health Organisation, American Cancer Society, World Cancer Research Fund International, and Cancer Research UK
- Yousaf-Khan U, van der Aalst C, de Jong PA, et al. Final screening round of the NELSON lung cancer screening trial: the effect of a 2.5-year screening interval. Thorax. 2017;72(1):48–56. doi:10.1136/thoraxjnl-2016-208655
- De Koning HJ et al. N Engl J Med. 2020. DOI:10.1056/ NEJMoa1911793
- De Koning HJ. 2018. Lung cancer CT-screening: NELSON mortality data. IASLC WCLC 2018. 09-25-18. Toronto, Canada.